
C.H. Rennie Hospital in Kakata, Margibi County in Liberia on March 4, 2015.
Photo © Dominic Chavez/World Bank
The 2014–2015 Ebola epidemic was the longest and deadliest Ebola epidemic in history, resulting in 28,616 cases and 11,310 deaths and reversing a decade of mortality and economic gains in Guinea, Liberia, and Sierra Leone. The statistics would probably have been very different, however, had an Ebola vaccine been available or even further along on the clinical development path.
This was not the first time that the world saw Ebola. It was first identified in 1976, but since all Ebola outbreaks prior to 2014-15 were relatively isolated, self-contained and of short duration, little was known about how to best manage large numbers of patients to improve survival, and there were no approved therapeutics or vaccines. Some medical countermeasures had been developed earlier and had been tested on animals. Robust testing of the safety of promising candidate vaccines only started in 2014, which means that efficacy testing in affected countries could only be initiated in early 2015, and the epidemic ended before most trials could be completed.
Moreover, most affected countries were overwhelmed, and were poorly equipped to support clinical research during the epidemic. Despite these shortcomings, data published in August 2015 suggested that one of the tested Ebola vaccine candidates was safe and effective. These results were confirmed in 2016 and the corresponding Ebola vaccine candidate is currently submitted for registration in the USA and in Europe.
A clear lesson that emerged from the Ebola experience is that research for medical countermeasures should be prioritized as part of epidemic preparedness, and that all countries need to develop strong and sustainable core capacities at the intersection of health systems and research. Public health, clinical care, and clinical research are important and interconnected components of a strong health system – and none can be divorced from the rest.
Recommendation 5 of the US National Academies of Sciences, Engineering, and Medicine report entitled “Integrating Clinical Research into Epidemic Response: The Ebola Experience” specifically states: “National governments should strengthen and incorporate research systems into their emergency preparedness and response systems for epidemic infectious diseases. The multilateral institutions (World Health Organization and the World Bank Group), and regional and international development agencies, and foundations working in global health should support national efforts by providing expertise and financing.”
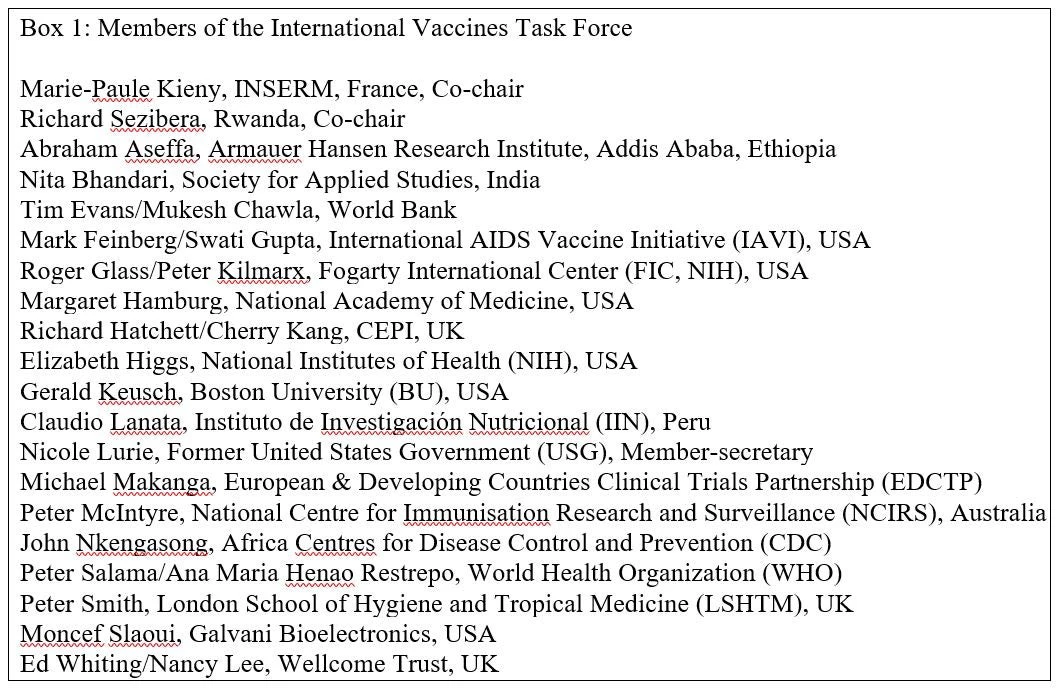
This task is fraught with challenges, not only because of competition across the economy and within the health sector for scarce resources, but also because of the incomplete understanding of the various options that countries and the global community have of strengthening research capacity in low-income countries. The first issue is discussed extensively in the report of the International Working Group on Financing Preparedness entitled “From Panic and Neglect to Investing in Health Security: Financing Pandemic Preparedness at a National Level,” which proposes ways in which national governments and development partners can finance investments in country and regional preparedness and response capacities for pandemics and other health emergencies. To address the second challenge – that of strengthening research capacity in low-income countries – the International Vaccines Task Force was created in October 2017 by the World Bank and the Coalition for Epidemic Preparedness Innovations (CEPI). (see Box 1 for a list of members).
A majority of the hot spots for emergence of new zoonotic pathogens (which comprise two-thirds of all new threats) are around communities that do not have the capacity to detect threats or respond to outbreaks, let alone conduct clinical research. The objective of the International Vaccines Task Force is to propose ways in which national governments, development partners, and the private sector can effectively and sustainably establish and finance clinical research capacity at the national and/or regional level. One of the objectives is that countries develop the capability to conduct clinical trials of vaccines, if and as needed.
The Task Force is looking at supporting the establishment of critical capacities, including appropriate physical infrastructure, trained health research workforce, functional ethics committees, regulatory capacity, expertise in social sciences, and basic public health capacity, such as surveillance and basic laboratory set-up in ways that are sustainable in the long run. For many countries, therefore, the starting points will be WHO’s R&D Blueprint for Action, which outlines how to improve research and development preparedness and response, focusing on a list of priority pathogens. These efforts should complement those already promoted under the Global Health Security Agenda (GHSA)and the Joint Evaluation Tool (JEE) which engage with stakeholders and partners’ initiatives to support country outbreak and health emergency preparedness. Both GHSA and JEE are already supported by the World Bank and the World Health Organization.
Taking vaccines from the pre-clinical to clinical research phases requires working in the reality of struggling health systems, where last-minute and ad-hoc capacity building is highly inefficient at the time of an outbreak. Efforts such as the Coalition for Epidemic Preparedness Innovations (CEPI) – which aim to move promising vaccine candidates through late pre-clinical studies in pre-epidemic periods and build technical platforms and institutional capacities that can be rapidly deployed against new and unknown pathogens – provide novel platforms for international collaboration where scientific preparedness can be undertaken to reduce time to creation and distribution of effective countermeasures.
The International Vaccines Task Force will deliver its final report in May 2018, in time for presentation at the World Health Assembly. We are open to ideas – and if you have thoughts and suggestions that we should consider on how best to strengthen national clinical research capacity, we would be delighted to hear them. Leave a comment, or email us at ivtf@worldbank.org.




Join the Conversation